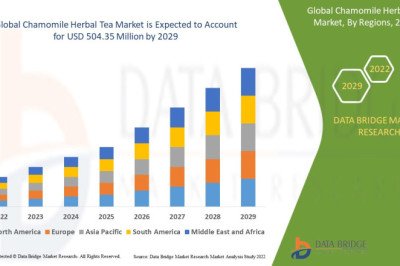
views
The terms lambda and kappa refer to the two distinct immunoglobulin light chain types found in mammals. However, there is no functional distinction between them that is currently understood, and both can happen with any of the five main categories of heavy chains. Each Immunoglobulin has either both or both of two identical light chains.
The ratios of and types differ between species and can be used to identify aberrant B cell clone growth. Some animals like sharks (Chondrichthyes) and bony fishes contain different types of light chains, such as the iota () chain (Teleostei). Antibodies typically have the same structure in most placental mammals. Jawed fish appear to be the most primitive creatures capable of producing antibodies equivalent to those found in mammals, despite the fact that many aspects of their adaptive immunity developed earlier.
The epitope of the antigen and the paratope of the Immunoglobulin interact. Different epitopes are typically organised discontinuously along the surface of an antigen, and dominant epitopes on a particular antigen are referred to be determinants. By spatial complementarity, antibodies and antigens interact (lock and key). The Fab-epitope contact is governed by weak, non-specific molecular forces, including as electrostatic forces, hydrogen bonds, hydrophobic interactions, and van der Waals forces. This indicates that the affinity of an Immunoglobulin towards an antigen is relative rather than absolute, and that the interaction between Immunoglobulin and antigen is reversible. An Immunoglobulin may cross-react with various antigens that have varying relative affinities as a result of relatively weak binding.
Read More - https://wordoid01.weebly.com/cmi-article/april-11th-2023